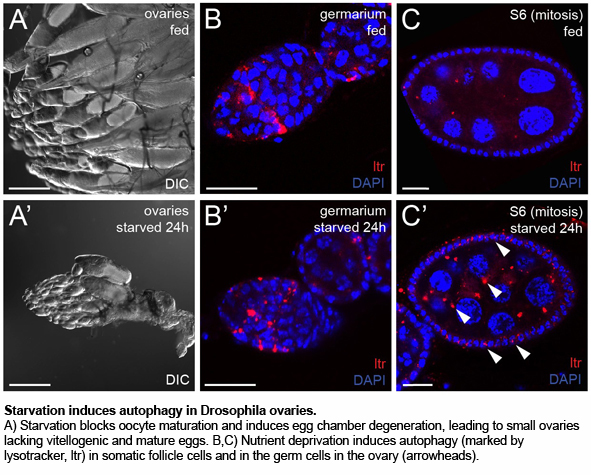
Case study: Drosophila melanogaster
Drosophila larvae have a storage organ called the fat body, which has similar functions to the liver and fat tissue in vertebrates. This storage unit acts as a reservoir for a large amount of potential energy. If an animal has just consumed food, then there is no need to utilise this energy store. Insulin is produced in response to this food, triggering a signalling cascade in the organism (Klionsky 2004). The enzyme “TOR”, a key element in this pathway, is involved in many regulatory events connected with energy metabolism, and also suppresses autophagy. It is now clear that this pathway provides a mechanism by which the insect’s cells can block autophagy if the organism has just fed (Klionsky 2004). However, this pathway also provides a means of inducing autophagy when the organism is starved. A lack of food/glucose leads to a lack of insulin, rendering the pathway inactive and making it necessary for the cell to tap into its storage reserves (Klionsky 2004). It was found that this ‘switch on’ for autophagy is developmentally programmed during the larval stage of Drosophila melanogaster; in other words it is programmed to occur. The hormone ecdysone appears to promote autophagy, and has been observed to increase in larva at set times during development (Klionsky 2004). This supports the theory that cell autophagy, as another form of cannibalism, is present in the insect Drosophila.